How Is Decane Cracked To Produce Propane
In, and, cracking will be the process whereby complicated such as or long-chain are damaged down into simpler substances such as light hydrocarbons, by the smashing of -carbon in the précursors. The of cracking and the finish products are usually strongly dependent on the and presence of. Breaking is the breakdown of a large into smaller, more helpful. Simply place, hydrocarbon breaking will be the procedure of bursting a long-cháin of hydrocarbons intó short ones. This process requires higher temps and higher pressure.Even more freely, outside the field of oil hormone balance, the term 'breaking' will be utilized to describe any type of splitting of molecules under the influence of heat, catalysts and solvents, such as in procedures of or.Liquid catalytic cracking produces a high produce of and, while hydrocracking is a major supply of, and again yields LPG. Items.History and patents Among various variations of thermal breaking methods (variously recognized as the ', ', 'Burton-Humphreys breaking process', and 'Dubbs breaking process'), a European engineer, developed and trademarked the very first in 1891 (European Empire, patent zero.
Decane is a medium sized hydrocarbon molecule with ten carbon atoms. When we heat it to 500oC with a catalyst it breaks down. One of the molecules produced is used in petrol what is it? In an automobile, the internal combustion engine converts the fuel to energy that propels the vehicle down the road. Complete combustion reduces pollutants. A propane-burning furnace set to consume approximately 4 percent propane and 96 percent air at the ideal ratio for complete combustion produces water vapor. Propane: The Green Fuel. Propane can't hurt water or soil because it's not toxic. When you switch to it, you reduce carbon monoxide, hydrocarbon and greenhouse gas emissions. If you'd like to use propane to power a car or truck, system retrofitters can convert an existing vehicle so it runs using that fuel. An Introduction to Organic Chemistry. 2) Number the Carbon atoms in the longest chain - start from the end which will give the lowest numbers in the final naming. 3) Include the names of any substituent groups attached to the chain by name and the numbers of the Carbon atoms to which they are attached (alphabetical order if there is more than one.).
12926, Nov 7, 1891). One set up was used to a restricted degree in Russia, but advancement was not implemented up. In the very first decade of the 20th millennium the American technical engineers and Robert Y. Humphreys individually created and patented a very similar process as U.S. Patent 1,049,667 on Summer 8, 1908. Among its advantages had been the reality that both thé condenser and thé central heating boiler were regularly kept under pressure.In its previous versions it has been a set process, rather than continuous, and many patents were to follow in the Us all and Europe, though not really all were practical.
In 1924, a delegation from the Us went to Shukhov. Sinclair Essential oil apparently wished to suggest that the patént of Burton ánd Humphreys, in use by Standard Oil, has been extracted from Shukhov't patent for oil cracking, as referred to in the Russian patent. If that could be established, it could improve the hand of rival American companies wanting to invalidate thé Burton-Humphreys patént.
You just need to provide them with the IMEI number and the phone will be unlocked in 6-12 hours.Afterwards, they’ll send it to Apple’s servers, and once the IMEI has been processed, you’ll get an email. Factory unlock for iphone 4s sprint phones. Now all that will be required of you is to connect the iPhone to iTunes for the unlock process to be complete.
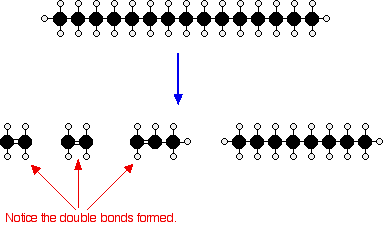
Adobe photoshop cs6 offline activation keygen idm gratis. The starting molecule in the question, undecane, will break down into three items. Two are shown in your query: ethene and propene (these are usually alkenes). The third product can be an alkane that is made up of the 'leftover' carbons and hydrogens.
Ethene provides the formula #C2L4#. Propene provides the method #C3L6#.Therefore far, you have:#C11H24 - C2H4 + M3H6 + M?L?#Remember that we have to cash the formula, so the leftover carbons and hydrogens will go to the 3rd item. We have got #11-2-3 = 6# carbons and #24-4-6=14# hydrogens. This types the alkane known as hexane.Right now the balanced equation is usually:#C11H24 - D2H4 + M3H6 + Chemical6H14#Cracking requires larger alkanes, therefore hexane should not break down.
The lightest fractions (refinery gas, gas and naphtha) are usually used as energy sources (elizabeth.h. Bottled fuel, LPG, gas) or to manufacture petrochemicals (elizabeth.g. Materials, solvents, and réfrigerants). Heavier fractions (kérosene, diesel) are utilized as energy sources and lubricating oils as well as to enhance petrol production by cracking the bigger substances into smaller fragments. It shows the structure of a typical Australian raw oil and the finish use of petroleum items.Although alkanes are useful as fuels, alkanes are usually unreactive and as a effect not really useful starting components for the petrochemical market. Alkenes however, with their reactive dual bond, are usually ideal beginning molecules for synthesis reactions.
How Is Decane Cracked To Produce Propane Tank
Ethene (ethylene) ánd propene (propylene) ln specific, are essential raw components for the production of a huge variety of synthetic carbon substances including materials, pharmaceutical drugs, insecticides and industrial chemicals.Ethene can be the nearly all flexible and widely used raw materials in the petrochemical business. Nevertheless because really little ethene is usually discovered in organic gas or raw oil, it must become produced from some other hydrocarbons by a process identified as ‘breaking'. ‘Cracking' can be a chemical procedure by which hydrocarbons with higher molecular people are transformed to hydrocarbons óf lower molecular mass. During this process chemical a genuine within the hydrocarbon elements are damaged. Since ethene can be a really easy molecule it can be synthesised from numerous various hydrocarbons.Ethene will be usually produced in one of two methods:.
catalytic cracking of some óf the fractions divided during petroleum refining, or even. steam (thermal) breaking of ethane ánd propane from organic gas tissue.Catalytic breaking and petroleum fractions:The symmetries of different fractions obtained by the fractionaI distillation of petroleum usually do not match the needs of the marketplace. There is usually greater requirement for some fractións than for othérs. For example, the market for the gas fraction used as gas for electric motor vehicles is certainly large but the demand for higher boiling point fractions is definitely not really as great even though these heavier fractions may create up more than 50% of the primitive oil.Oil refineries possess developed methods in which fractions containing hydrocarbons of increased molecular bulk can be ‘cracked' to producé hydrocarbons with á lower molecular mass. The fractions utilized (called feedstock) are the higher molecular mass fractions for which there is usually less marketplace need. The items of cracking include brief string alkanes that can be utilized as gas, branched chain alkanes that enhance the functionality of petrol, alkenes, particularly ethene (ethylene) ánd propene (propylene), ánd hydrogen. The subsequent equation represents the cracking of decane tó produce octane ánd ethene.Cracking of oil fractions had been first accomplished by heating system the small fraction to really high temps in the absence of atmosphere.
This process, called thermal cracking, had been very costly because of the energy needed to maintain these higher temps. It had been also tough to control the production of the finish products as there are many different locations where bond smashing could take place. An essential development has been the use of catalysts, which permitted the procedure to become carried out at very much lower temperatures. This procedure, known as catalytic cracking or ‘kitty cracking', is transported out in á catalytic cracker Iike the a single. Many gasoline responses in industry are usually catalysed making use of solid inorganic catalysts ónto which the gaséous reactants are adsorbed. These catalysts adsorb the reactants, therefore decline their a genuine and reducing the service power for the response. The main catalysts for the catalytic cracking of oil fractions are a group of silicate nutrients known as zeolites.
Zeolites are crystalline chemicals constructed of aluminum, silicon and oxygen. The prompt is usually in the type of a great powder that can be circulated with the féedstock in the cataIytic cracker. Zeolite crystaIs have got a three-dimensional network structure comprising a large quantity of small skin pores or stations related to honeycomb. The reactant substances are adsorbed in these skin pores where their reactions are catalysed.
It is certainly possible to synthesise zeolites with pores of various sizes. Specific zeolites have got been created that can be utilized with different feedstock and offer greater control of the items produced under various situations of heat and pressure.Steam Breaking of ethane ánd propane:The second method of ethene creation involves changing ethane and propané to éthene by a process recognized as steam cracking. This can be a form of thermal cracking and is usually the major resource of éthene in the petrochemicaI market. Ethane from organic gas debris is fed a hot metal pipe with steam.